Mercury »
PDB 12ca-1czm »
1cni »
Mercury in PDB 1cni: X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site
Enzymatic activity of X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site
All present enzymatic activity of X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site:
4.2.1.1;
4.2.1.1;
Protein crystallography data
The structure of X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site, PDB code: 1cni
was solved by
C.A.Lesburg,
D.W.Christianson,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 8.00 / 1.80 |
| Space group | P 1 21 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 42.700, 41.700, 73.000, 90.00, 104.60, 90.00 |
| R / Rfree (%) | 16.1 / n/a |
Other elements in 1cni:
The structure of X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site also contains other interesting chemical elements:
| Zinc | (Zn) | 1 atom |
Mercury Binding Sites:
The binding sites of Mercury atom in the X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site
(pdb code 1cni). This binding sites where shown within
5.0 Angstroms radius around Mercury atom.
In total only one binding site of Mercury was determined in the X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site, PDB code: 1cni:
In total only one binding site of Mercury was determined in the X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site, PDB code: 1cni:
Mercury binding site 1 out of 1 in 1cni
Go back to
Mercury binding site 1 out
of 1 in the X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site
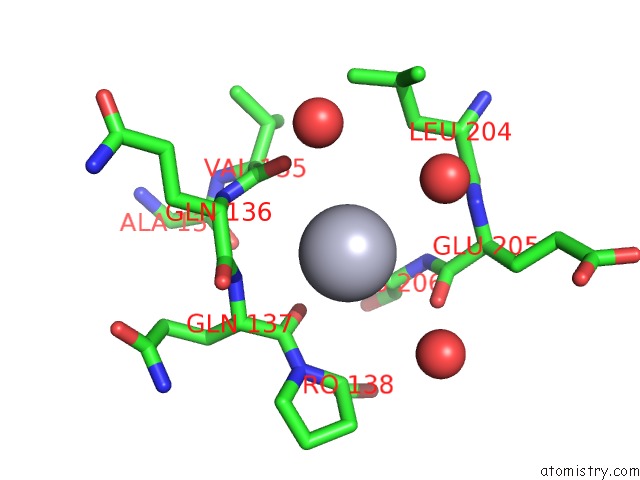
Mono view
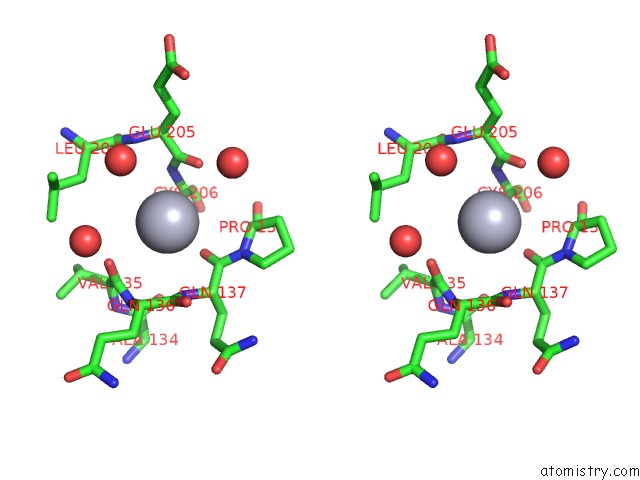
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 1 of X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site within 5.0Å range:
|
Reference:
C.A.Lesburg,
D.W.Christianson.
X-Ray Crystallographic Studies of Engineered Hydrogen Bond Networks in A Protein-Zinc Binding Site J.Am.Chem.Soc. V. 117 6838 1995.
ISSN: ISSN 0002-7863
Page generated: Fri Aug 8 08:55:06 2025
ISSN: ISSN 0002-7863
Last articles
K in 2IHUK in 2IH3
K in 2IH1
K in 2HZV
K in 2IEH
K in 2IBY
K in 2IBW
K in 2I2X
K in 2IB8
K in 2IB9