Mercury »
PDB 2o1g-3b4f »
2yaw »
Mercury in PDB 2yaw: Hg Inhibited Sulfur Oxygenase Reductase
Enzymatic activity of Hg Inhibited Sulfur Oxygenase Reductase
All present enzymatic activity of Hg Inhibited Sulfur Oxygenase Reductase:
1.13.11.55;
1.13.11.55;
Protein crystallography data
The structure of Hg Inhibited Sulfur Oxygenase Reductase, PDB code: 2yaw
was solved by
A.Veith,
T.Urich,
K.Seyfarth,
J.Protze,
C.Frazao,
A.Kletzin,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 49.04 / 2.50 |
| Space group | I 4 |
| Cell size a, b, c (Å), α, β, γ (°) | 161.876, 161.876, 154.373, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 16.2 / 18.3 |
Other elements in 2yaw:
The structure of Hg Inhibited Sulfur Oxygenase Reductase also contains other interesting chemical elements:
| Iron | (Fe) | 6 atoms |
Mercury Binding Sites:
Pages:
>>> Page 1 <<< Page 2, Binding sites: 11 - 12;Binding sites:
The binding sites of Mercury atom in the Hg Inhibited Sulfur Oxygenase Reductase (pdb code 2yaw). This binding sites where shown within 5.0 Angstroms radius around Mercury atom.In total 12 binding sites of Mercury where determined in the Hg Inhibited Sulfur Oxygenase Reductase, PDB code: 2yaw:
Jump to Mercury binding site number: 1; 2; 3; 4; 5; 6; 7; 8; 9; 10;
Mercury binding site 1 out of 12 in 2yaw
Go back to
Mercury binding site 1 out
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
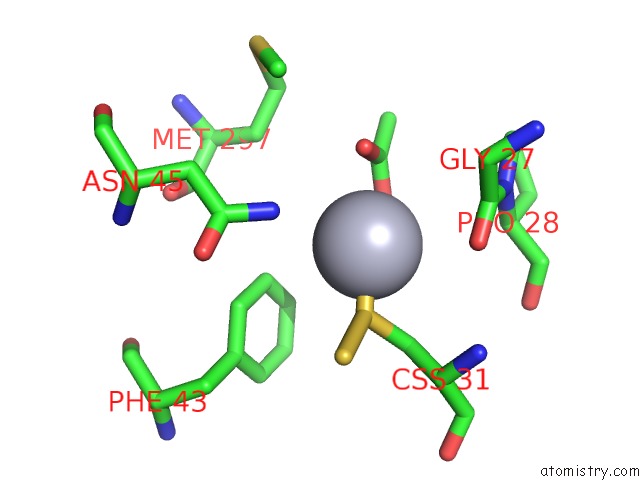
Mono view
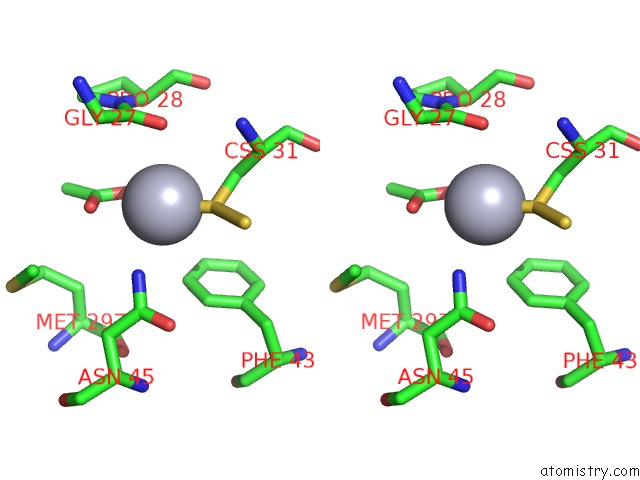
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 1 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
Mercury binding site 2 out of 12 in 2yaw
Go back to
Mercury binding site 2 out
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
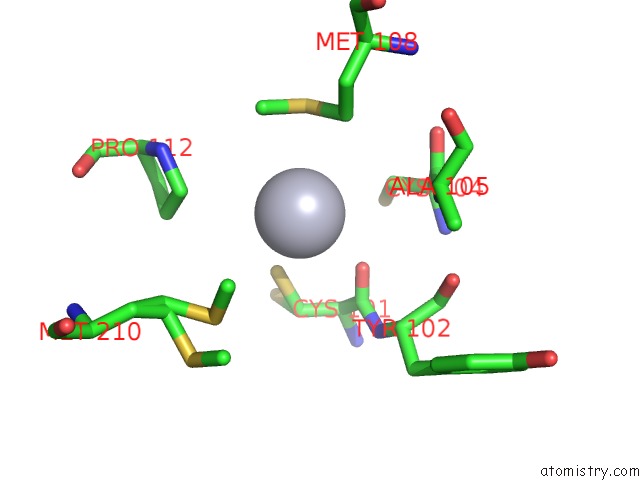
Mono view
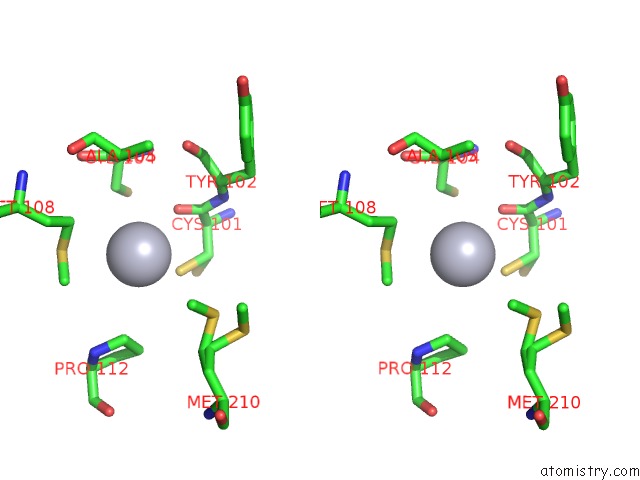
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 2 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
Mercury binding site 3 out of 12 in 2yaw
Go back to
Mercury binding site 3 out
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
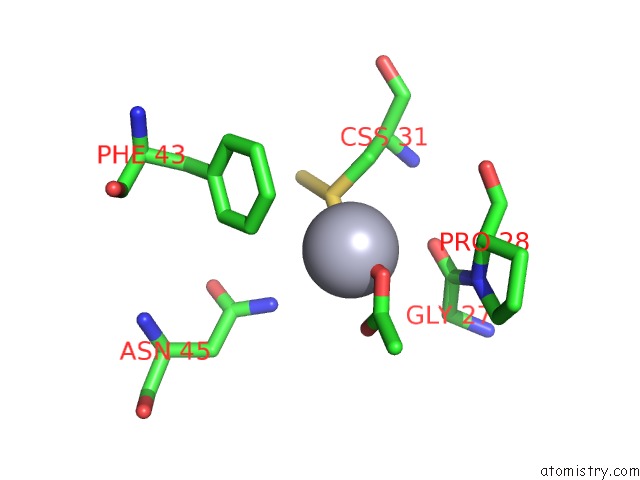
Mono view
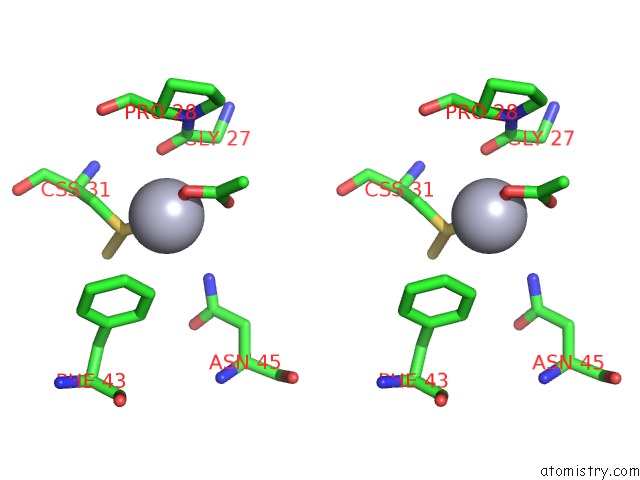
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 3 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
Mercury binding site 4 out of 12 in 2yaw
Go back to
Mercury binding site 4 out
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
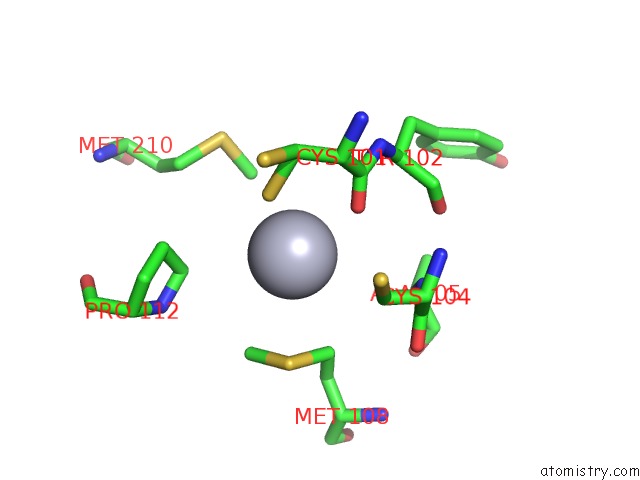
Mono view
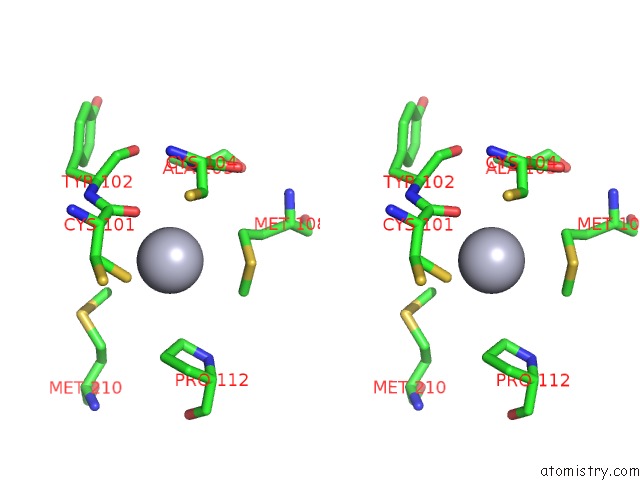
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 4 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
Mercury binding site 5 out of 12 in 2yaw
Go back to
Mercury binding site 5 out
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
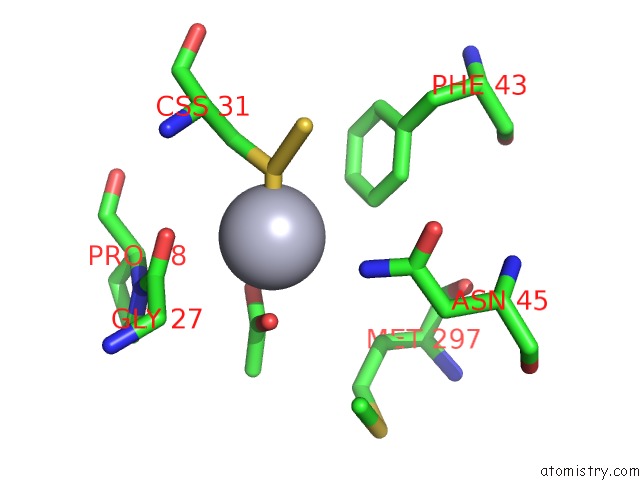
Mono view
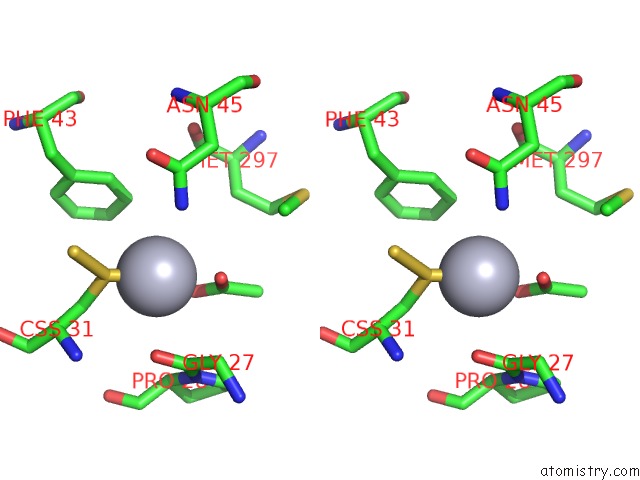
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 5 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
Mercury binding site 6 out of 12 in 2yaw
Go back to
Mercury binding site 6 out
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
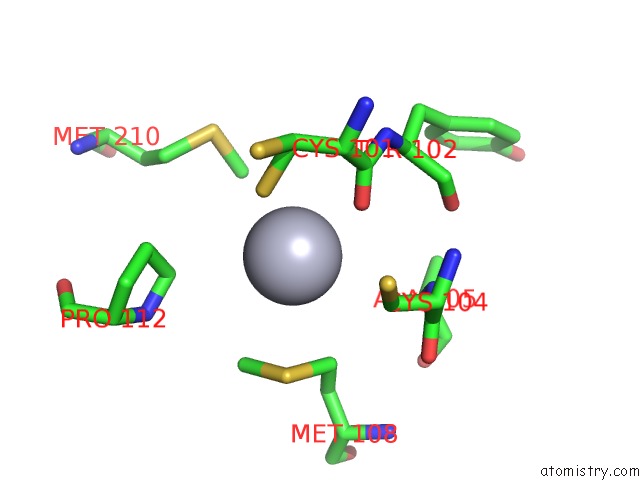
Mono view
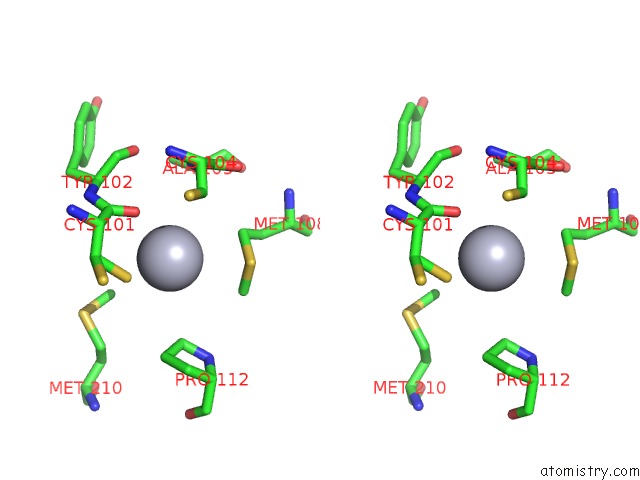
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 6 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
Mercury binding site 7 out of 12 in 2yaw
Go back to
Mercury binding site 7 out
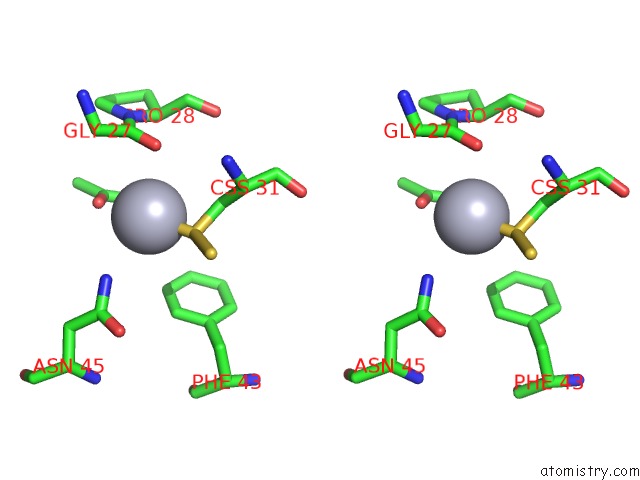
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
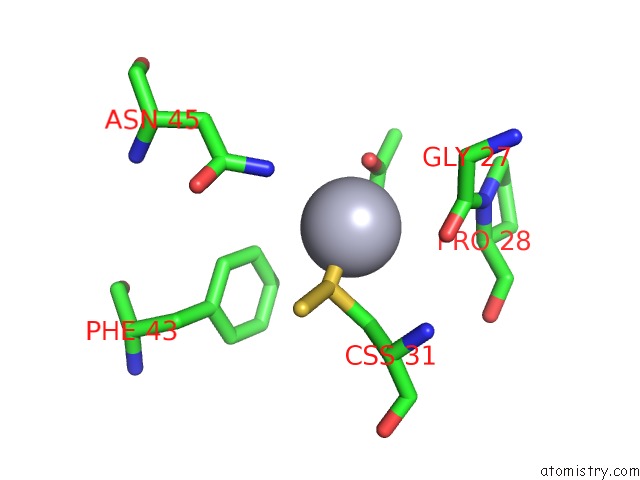
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 7 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
Mercury binding site 8 out of 12 in 2yaw
Go back to
Mercury binding site 8 out
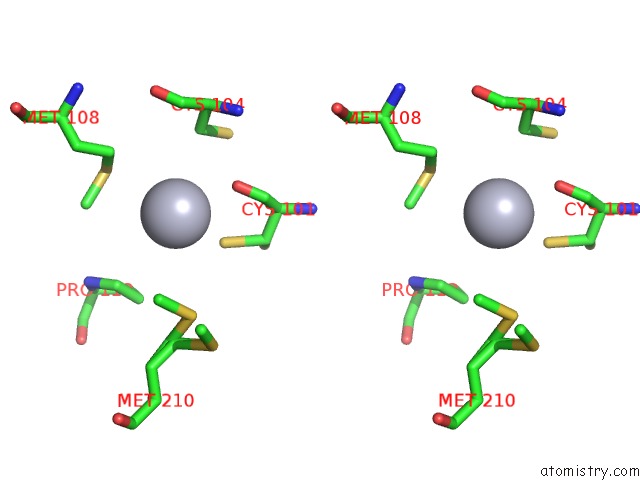
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
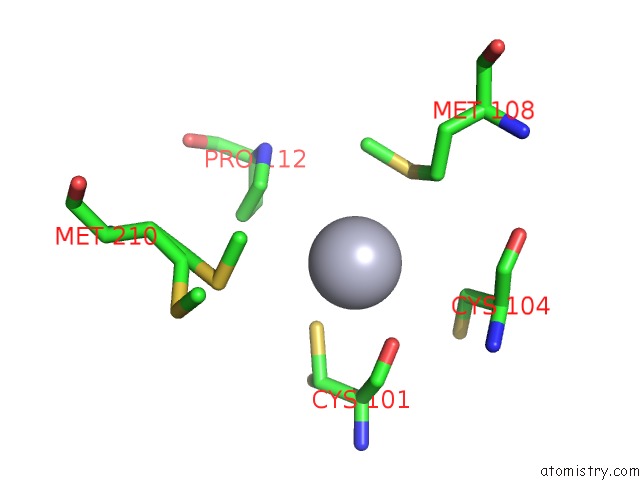
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 8 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
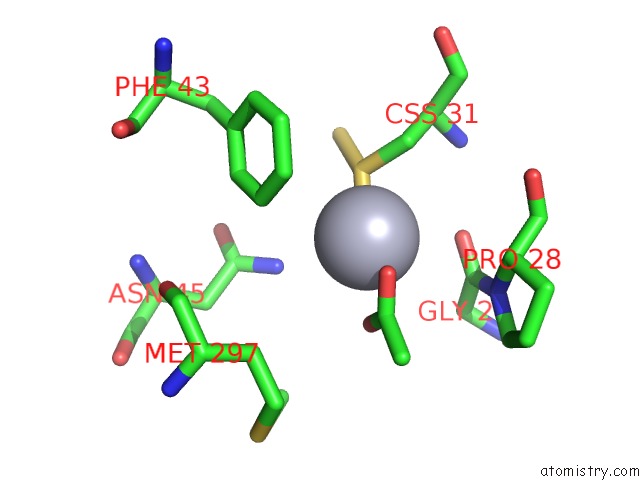
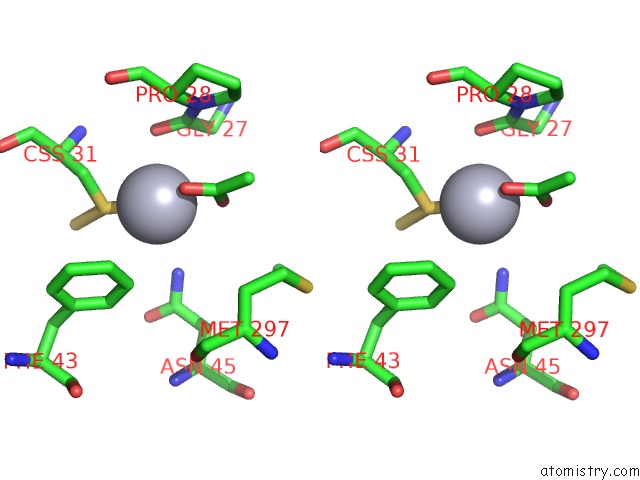
Mercury binding site 9 out of 12 in 2yaw
Go back to
Mercury binding site 9 out
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 9 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
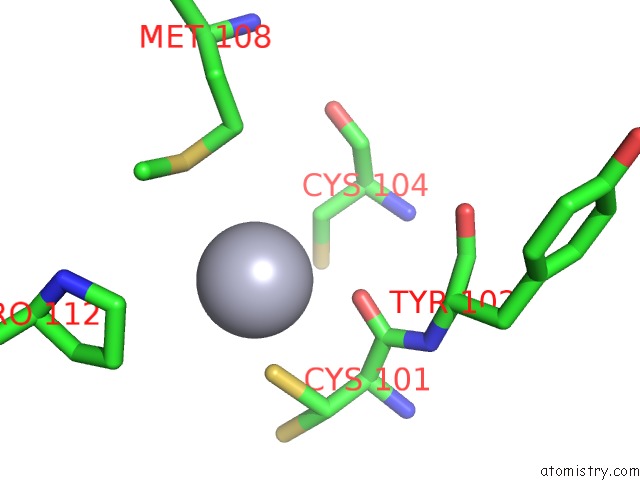
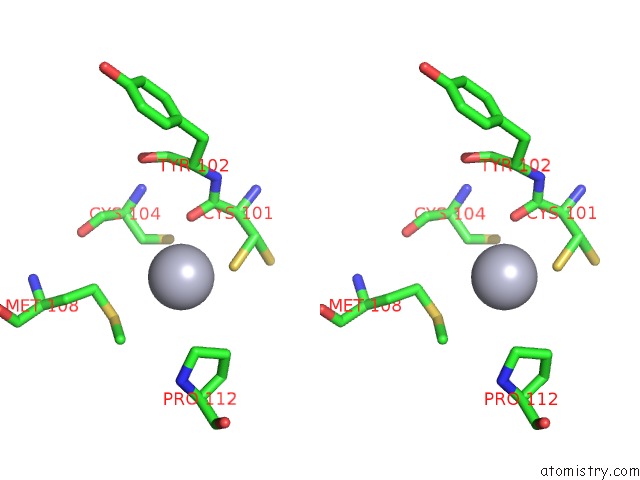
Mercury binding site 10 out of 12 in 2yaw
Go back to
Mercury binding site 10 out
of 12 in the Hg Inhibited Sulfur Oxygenase Reductase
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 10 of Hg Inhibited Sulfur Oxygenase Reductase within 5.0Å range:
|
Reference:
A.Veith,
T.Urich,
K.Seyfarth,
J.Protze,
C.Frazao,
A.Kletzin.
Substrate Pathways and Mechanisms of Inhibition in the Sulfur Oxygenase Reductase of Acidianus Ambivalens. Front.Microbiol. V. 2 37 2011.
ISSN: ESSN 1664-302X
PubMed: 21747782
DOI: 10.3389/FMICB.2011.00037
Page generated: Sun Aug 11 03:06:12 2024
ISSN: ESSN 1664-302X
PubMed: 21747782
DOI: 10.3389/FMICB.2011.00037
Last articles
Ca in 5S5XCa in 5S5Y
Ca in 5S5W
Ca in 5S5V
Ca in 5S5U
Ca in 5S5T
Ca in 5S5S
Ca in 5S5R
Ca in 5S5Q
Ca in 5S5P