Mercury »
PDB 3kbc-3wa8 »
3m14 »
Mercury in PDB 3m14: Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor
Enzymatic activity of Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor
All present enzymatic activity of Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor:
4.2.1.1;
4.2.1.1;
Protein crystallography data
The structure of Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor, PDB code: 3m14
was solved by
J.Schulze Wischeler,
A.Heine,
G.Klebe,
N.U.Sandner,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 10.00 / 1.38 |
| Space group | P 1 21 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 42.138, 41.349, 72.033, 90.00, 104.30, 90.00 |
| R / Rfree (%) | 12.7 / 17.6 |
Other elements in 3m14:
The structure of Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor also contains other interesting chemical elements:
| Zinc | (Zn) | 1 atom |
Mercury Binding Sites:
The binding sites of Mercury atom in the Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor
(pdb code 3m14). This binding sites where shown within
5.0 Angstroms radius around Mercury atom.
In total only one binding site of Mercury was determined in the Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor, PDB code: 3m14:
In total only one binding site of Mercury was determined in the Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor, PDB code: 3m14:
Mercury binding site 1 out of 1 in 3m14
Go back to
Mercury binding site 1 out
of 1 in the Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor
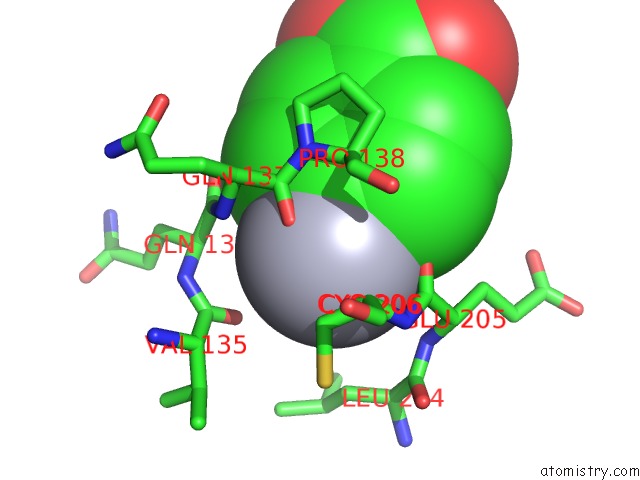
Mono view
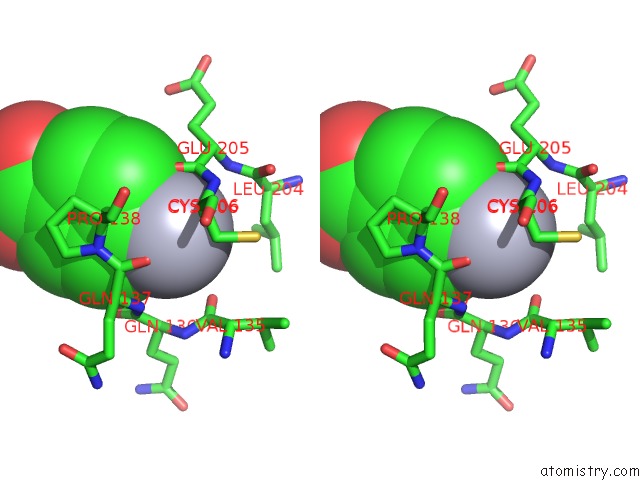
Stereo pair view

Mono view

Stereo pair view
A full contact list of Mercury with other atoms in the Hg binding
site number 1 of Carbonic Anhydrase II in Complex with Novel Sulfonamide Inhibitor within 5.0Å range:
|
Reference:
J.Schulze Wischeler,
N.U.Sandner,
M.Haake,
C.Supuran,
A.Heine,
G.Klebe.
Structural Investigation and Inhibitor Studies on Carbonic Anhydrase II To Be Published.
Page generated: Fri Aug 8 10:13:43 2025
Last articles
I in 2WABI in 2VMA
I in 2WK6
I in 2W86
I in 2W7Y
I in 2VUW
I in 2VLW
I in 2VGZ
I in 2VA7
I in 2VG7